
Workshop 2 - Training course in data analysis for genomic surveillance of African malaria vectors
Module 3 - Detecting CNVs from genome sequence data#
Theme: Data
In this module we will learn about how we detect copy number variants (CNVs) from whole-genome sequencing data, and how to access and visualise the individual-level CNV data using the malariagen_data package in Python.
Learning objectives#
At the end of this module you will be able to:
Explain what copy number variation is, and what is meant by CNV alleles
Explain how we detect CNVs in whole-genome sequence data from MalariaGEN
Access and visualise MalariaGEN CNV data
Lecture#
English#
Français#
Please note that the code in the cells below might differ from that shown in the video. This can happen because Python packages and their dependencies change due to updates, necessitating tweaks to the code.
Introduction: what are CNVs?#
Copy number variants (CNVs) are a type of mutation that changes the number of copies of a genetic region on the chromosome. That region might be deleted (reduction in copy number, called a deletion), or might be copied (increase in copy number, called a duplication or amplification). When considering the evolution of insecticide resistance, we tend to focus on increases in copy number, because they could lead to increased expression of genes that metabolise insecticides. But this doesn’t mean that it is impossible for deletions to also play an important role.
In the figure below, an example “normal” sequence (top line) is shown with the sequence in blue either deleted (middle line) or duplicated (bottom line). This simple example involves a CNV of just 11 base pairs, but they can be as long as hundreds of thousands of bp, and the analysis performed by MalariaGEN looks for CNVs on the scale of several hundreds of bp at least.
A normal genetic region that is not repeated through the genome should typically be found in two copies in each cell of a mosquito. This is because mosquitoes are diploid, and therefore carry two copies of each chromosome (except for sex chromosomes in males). Therefore, the “normal” copy number for a given genetic region is 2. We consider that there is a CNV in a region if we find samples with copy number greater or less than two. For example, the following copy numbers might be found in samples with or without duplications in a genetic region.
A CNV can sometimes include one or more gene-coding regions. Since multiple genes can be affected by a single CNV, it can sometimes be difficult to determine the mechanistic link between the CNV and any phenotypic effect that it is found to be associated with. In the figure below, a CNV creates two full copies of genes X and Y, and a partial copy of gene Z (which is unlikely to create a functional gene).
As described in the video lecture, we detect CNVs in MalariaGEN using whole genome sequencing data through a combination of coverage-based analysis (i.e., counting how much of the sequencing data comes from each region in the genome) and examination of reads from the CNV breakpoints (discordant reads).
Let’s now look at how we access CNV data from MalariaGEN.
Setup#
First we need to install and import some Python packages.
%pip install -q --no-warn-conflicts malariagen_data
import warnings
warnings.simplefilter(action='ignore', category=UserWarning)
import malariagen_data
import numpy as np
import pandas as pd
import re
import seaborn as sns
import plotly.io as pio
pio.renderers.default = "notebook+colab"
import matplotlib.pyplot as plt
%matplotlib inline
import bokeh.io as bkio
bkio.output_notebook()
#import os
#os.environ["MGEN_SHOW_PROGRESS"] = "0"
Once the packages are imported, set up data access through the malariagen_data package. Note that authentication is required to access data through the package, please follow the instructions here.
ag3 = malariagen_data.Ag3()
ag3
| MalariaGEN Ag3 API client | |
|---|---|
| Please note that data are subject to terms of use, for more information see the MalariaGEN website or contact support@malariagen.net. See also the Ag3 API docs. | |
| Storage URL | gs://vo_agam_release_master_us_central1 |
| Data releases available | 3.0, 3.1, 3.2, 3.3, 3.4, 3.5, 3.6, 3.7, 3.8, 3.9, 3.10, 3.11, 3.12, 3.13, 3.14 |
| Results cache | None |
| Cohorts analysis | 20250131 |
| AIM analysis | 20220528 |
| Site filters analysis | dt_20200416 |
| Software version | malariagen_data 15.0.1 |
| Client location | Iowa, United States (Google Cloud us-central1) |
Inspect which sample sets are available within the MalariaGEN Ag3.0 data release.
df_sample_sets = ag3.sample_sets(release="3.0")
df_sample_sets[['sample_set','sample_count','study_id']]
| sample_set | sample_count | study_id | |
|---|---|---|---|
| 0 | AG1000G-AO | 81 | AG1000G-AO |
| 1 | AG1000G-BF-A | 181 | AG1000G-BF-1 |
| 2 | AG1000G-BF-B | 102 | AG1000G-BF-1 |
| 3 | AG1000G-BF-C | 13 | AG1000G-BF-2 |
| 4 | AG1000G-CD | 76 | AG1000G-CD |
| 5 | AG1000G-CF | 73 | AG1000G-CF |
| 6 | AG1000G-CI | 80 | AG1000G-CI |
| 7 | AG1000G-CM-A | 303 | AG1000G-CM-1 |
| 8 | AG1000G-CM-B | 97 | AG1000G-CM-2 |
| 9 | AG1000G-CM-C | 44 | AG1000G-CM-3 |
| 10 | AG1000G-FR | 23 | AG1000G-FR |
| 11 | AG1000G-GA-A | 69 | AG1000G-GA-1 |
| 12 | AG1000G-GH | 100 | AG1000G-GH |
| 13 | AG1000G-GM-A | 74 | AG1000G-GM-1 |
| 14 | AG1000G-GM-B | 31 | AG1000G-GM-2 |
| 15 | AG1000G-GM-C | 174 | AG1000G-GM-3 |
| 16 | AG1000G-GN-A | 45 | AG1000G-GN-ML |
| 17 | AG1000G-GN-B | 185 | AG1000G-GN-ML |
| 18 | AG1000G-GQ | 10 | AG1000G-GQ |
| 19 | AG1000G-GW | 101 | AG1000G-GW |
| 20 | AG1000G-KE | 86 | AG1000G-KE |
| 21 | AG1000G-ML-A | 60 | AG1000G-ML-1 |
| 22 | AG1000G-ML-B | 71 | AG1000G-ML-2 |
| 23 | AG1000G-MW | 41 | AG1000G-MW |
| 24 | AG1000G-MZ | 74 | AG1000G-MZ |
| 25 | AG1000G-TZ | 300 | AG1000G-TZ |
| 26 | AG1000G-UG | 290 | AG1000G-UG |
| 27 | AG1000G-X | 669 | AG1000G-X |
In this module we will use the Côte d’Ivoire sample set (“AG1000G-CI”) from the Ag3.0 release to demonstrate accessing CNV data.
Load the CNV HMM data#
The HMM data include:
The raw coverage calls (“call_RawCov” - the number of reads aligning in each 300bp window along the chromosome).
Normalised coverage calls (“call_NormCov” - the raw coverage divided by expected coverage and multiplied by 2, such unduplicated regions have the expected diploid copy number value of 2).
Copy number calls, which is the output of the HMM (“call_CN” - the estimated copy number in each 300 bp window).
hmm = ag3.cnv_hmm(region="2R", sample_sets="AG1000G-CI").set_index(samples="sample_id")
hmm
<xarray.Dataset> Size: 144MB
Dimensions: (variants: 205151, samples: 77)
Coordinates:
variant_position (variants) int32 821kB dask.array<chunksize=(65536,), meta=np.ndarray>
variant_end (variants) int32 821kB dask.array<chunksize=(65536,), meta=np.ndarray>
variant_contig (variants) uint8 205kB dask.array<chunksize=(205151,), meta=np.ndarray>
* samples (samples) object 616B 'AY0072-C' ... 'AY0058-C'
Dimensions without coordinates: variants
Data variables:
call_CN (variants, samples) int8 16MB dask.array<chunksize=(65536, 40), meta=np.ndarray>
call_RawCov (variants, samples) int32 63MB dask.array<chunksize=(65536, 40), meta=np.ndarray>
call_NormCov (variants, samples) float32 63MB dask.array<chunksize=(65536, 40), meta=np.ndarray>
sample_coverage_variance (samples) float32 308B dask.array<chunksize=(77,), meta=np.ndarray>
sample_is_high_variance (samples) bool 77B dask.array<chunksize=(77,), meta=np.ndarray>
Attributes:
contigs: ('2R', '2L', '3R', '3L', 'X')By inspecting the dataset, we can see that the data comprise 77 samples and 205,151 variants. Here each “variant” is one 300bp window, so the first value will represent the first 300 bp window, the second value will be the second window, and so on.
As an example in this workbook, we will look at the coverage data in four samples.
sample_names = ['AY0046-C', 'AY0062-C', 'AY0055-C', 'AY0034-C']
We can plot coverage in the Cyp6aa / Cyp6p cluster of genes, which have been shown to be involved in insecticide resistance.
The plots below show this region of the genome, which is on chromosome 2R between positions 28,460,000–28,580,000, with genomic position on the X axis. The black markers show normalised coverage, while the blue line shows the estimated copy number (i.e., the output of the HMM) at each 300 bp window.
for s in sample_names:
ag3.plot_cnv_hmm_coverage(s, sample_set='AG1000G-CI', region='2R:28,460,000-28,580,000')
From the plots above, we can see that each of those four samples have increased copy number (blue line higher than 2) in at least part of the region which we are looking at. Samples 2 and 3 look like they each have two CNVs in the region (two areas where copy number increases and then drops again).
The malariagen package also provides a convenient function to visualise the HMM output for many samples at once, as a heatmap.
ag3.plot_cnv_hmm_heatmap(
region='2R:28,460,000-28,580,000',
sample_sets='AG1000G-CI',
row_height=5
);
We can now look more precisely at which CNV alleles those two samples carry by looking at the discordant read data.
Load the CNV discordant read calls#
The CNV discordant read calls provide allele-specific CNV genotypes at a selection of genomic loci known to be involved in insecticide resistance.
discordant_read_calls = (
ag3
.cnv_discordant_read_calls(contig="2R", sample_sets="AG1000G-CI")
.set_index(samples="sample_id", variants="variant_id")
)
discordant_read_calls
<xarray.Dataset> Size: 6kB
Dimensions: (variants: 47, samples: 80)
Coordinates:
variant_position (variants) int32 188B dask.array<chunksize=(47,), meta=np.ndarray>
variant_end (variants) int32 188B dask.array<chunksize=(47,), meta=np.ndarray>
variant_contig (variants) uint8 47B dask.array<chunksize=(47,), meta=np.ndarray>
* samples (samples) object 640B 'AY0072-C' ... 'AY00...
* variants (variants) object 376B 'Ace1_Dup0' ... 'Cy...
Data variables:
variant_Region (variants) object 376B dask.array<chunksize=(47,), meta=np.ndarray>
variant_StartBreakpointMethod (variants) int32 188B dask.array<chunksize=(47,), meta=np.ndarray>
variant_EndBreakpointMethod (variants) int32 188B dask.array<chunksize=(47,), meta=np.ndarray>
call_genotype (variants, samples) int8 4kB dask.array<chunksize=(47, 64), meta=np.ndarray>
sample_coverage_variance (samples) float32 320B dask.array<chunksize=(80,), meta=np.ndarray>
sample_is_high_variance (samples) bool 80B dask.array<chunksize=(80,), meta=np.ndarray>
Attributes:
contigs: ('2R', '2L', '3R', '3L', 'X')Let’s pull out the discordant read calls in the Cyp6aa / Cyp6p region for the four samples we focused on above.
# Locate the CNV alleles from the Cyp6aa / Cyp6p region
loc_cyp6aap = discordant_read_calls["variant_Region"].values == 'Cyp6aa/Cyp6p'
# Select data for our genes and samples of interest
discordant_read_calls_cyp6aap = (
discordant_read_calls
.isel(variants=loc_cyp6aap)
.sel(samples=sample_names)
)
# Convert genotypes to a pandas DataFrame
df_cyp6aap = discordant_read_calls_cyp6aap["call_genotype"].to_pandas()
# Remove CNVs that are absent in all samples
df_cyp6aap = df_cyp6aap.loc[(df_cyp6aap != 0).any(axis=1)]
df_cyp6aap
| samples | AY0046-C | AY0062-C | AY0055-C | AY0034-C |
|---|---|---|---|---|
| variants | ||||
| Cyp6aap_Dup0 | 0 | 1 | 1 | 1 |
| Cyp6aap_Dup7 | 0 | 1 | 1 | 0 |
| Cyp6aap_Dup11 | 0 | 0 | 1 | 0 |
| Cyp6aap_Dup15 | 0 | 1 | 0 | 1 |
Each of the CNV alleles in this region are labelled “Cyp6aap_Dupxx”, where xx is an identifier associated with each allele. “Dup0” is a special name which is not associated with a given allele, but instead indicates whether any gene in the region carried a CNV based on modal copy number (i.e., whether the HMM output indicated a CNV in this gene).
From the table above, we can see that sample AY0046-C has no CNV, AY0034-C has one CNV, and samples 2 and 3 have two CNVs each. AY0062-C has Dup7 and Dup15, while AY0055-C has Dup7 and Dup11.
Let’s check where each of these CNVs start and end, and see whether this corresponds to the coverage increases we saw in the HMM data.
dup_positions = (
discordant_read_calls
.reset_coords()
[["variant_position", "variant_end"]]
.sel(variants=["Cyp6aap_Dup7", "Cyp6aap_Dup11", "Cyp6aap_Dup15"])
.to_dataframe()
)
dup_positions
| variant_position | variant_end | |
|---|---|---|
| variants | ||
| Cyp6aap_Dup7 | 28478057 | 28486036 |
| Cyp6aap_Dup11 | 28487546 | 28518123 |
| Cyp6aap_Dup15 | 28465673 | 28555300 |
Load the per-gene modal copy number data#
We can also look at individual genes and ask whether copy number was increased in each of those genes. This table shows the copy number observed for each gene in the Cyp6aa and Cyp6p cluster, with 2 being the normal diploid copy number (no CNV).
# Access per-gene modal copy number data for all genes in chromosome arm 2R
gene_copy_number = (
ag3.gene_cnv(region="2R", sample_sets="AG1000G-CI")
.set_index(genes="gene_id", samples="sample_id")
)
# Define genes of interest
cyp6aap_genes = dict(AGAP002862='Cyp6aa1',
AGAP013128='Cyp6aa2',
AGAP002868='Cyp6p1',
AGAP002869='Cyp6p2',
AGAP002865='Cyp6p3',
AGAP002867='Cyp6p4',
AGAP002866='Cyp6p5')
# Select data for genes of interest, excluding samples with poor quality HMM data
cyp6aap_gene_copy_number = (
gene_copy_number["CN_mode"]
.sel(genes=list(cyp6aap_genes.keys()))
.where(~gene_copy_number.sample_is_high_variance, drop=True)
.transpose()
.to_pandas()
.rename(cyp6aap_genes, axis='columns')
)
cyp6aap_gene_copy_number
| genes | Cyp6aa1 | Cyp6aa2 | Cyp6p1 | Cyp6p2 | Cyp6p3 | Cyp6p4 | Cyp6p5 |
|---|---|---|---|---|---|---|---|
| samples | |||||||
| AY0072-C | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 |
| AY0062-C | 4.0 | 4.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 |
| AY0055-C | 3.0 | 3.0 | 4.0 | 4.0 | 4.0 | 4.0 | 4.0 |
| AY0034-C | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 |
| AY0078-C | 3.0 | 3.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 |
| ... | ... | ... | ... | ... | ... | ... | ... |
| AY0017-C | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 |
| AY0043-C | 4.0 | 4.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 |
| AY0013-C | 4.0 | 4.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 |
| AY0036-C | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 |
| AY0058-C | 2.0 | 2.0 | 4.0 | 4.0 | 4.0 | 4.0 | 4.0 |
77 rows × 7 columns
We can show this as a heatmap for easier viewing. We’ll just show the top 25 samples to keep the table small.
def plot_gene_cnv_heatmap(data):
# create a figure
figsize = data.shape[1], data.shape[0]*0.7
fig, ax = plt.subplots(figsize=figsize)
# plot a heatmap
sns.heatmap(
data,
annot=True,
cbar=False,
cmap='bwr',
center=2,
linewidths=0.1,
ax=ax
)
# tidy plot
ax.tick_params(
axis='both',
which='major',
labelsize=10,
labelbottom=False,
bottom=False,
top=False,
labeltop=True,
left=False
)
ax.xaxis.set_label_position('top')
plot_gene_cnv_heatmap(cyp6aap_gene_copy_number.iloc[:25])
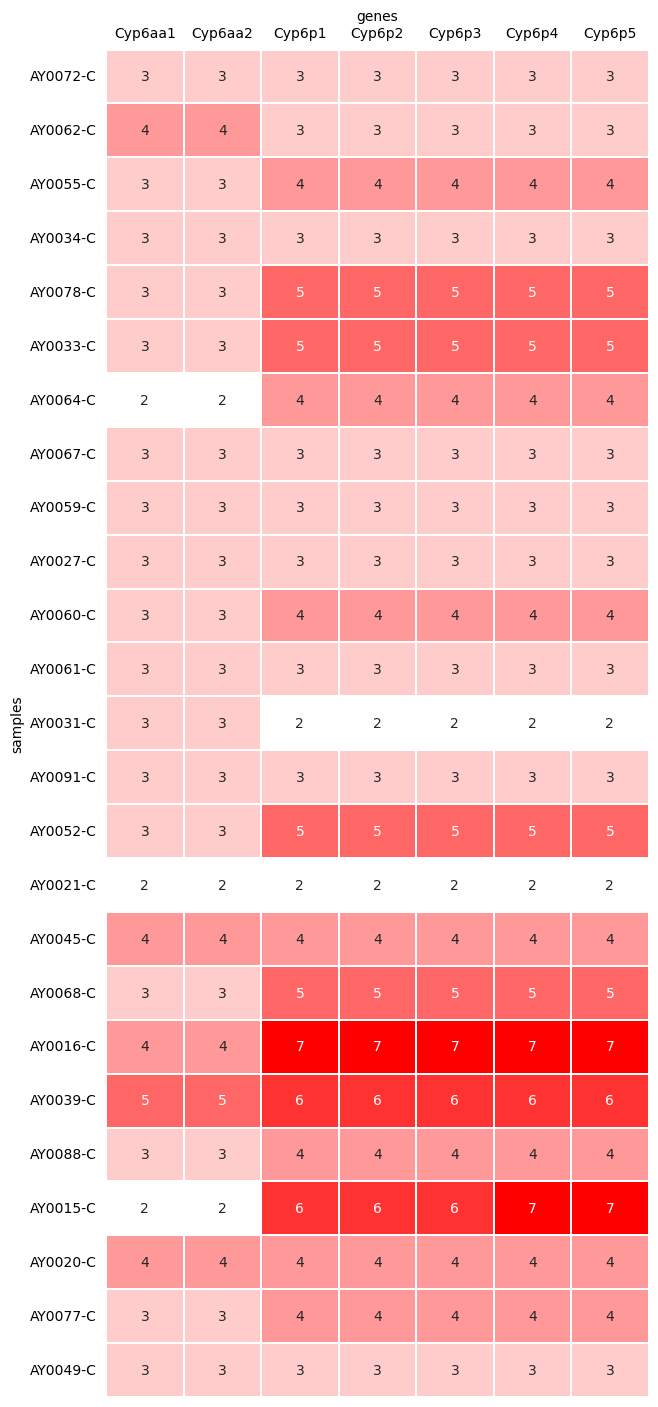
Notice how the copy number for each gene in each sample corresponds to the HMM in that gene’s region in your coverage plots above.
Exercises#
English#
Open this notebook in Google Colab and run it for yourself from top to bottom. Hint: click the rocket icon () at the top of the page, then select “Colab” from the drop-down menu. When colab opens, click the “Edit” menu, then select “Clear all outputs”, then begin running the cells. Make sure it runs without any errors, and take some time to try and understand what is being shown in the output.
Now go back to the “Load the CNV HMM data” section. Change the code so that it looks at chromosome 3R from position 28,570,000 to 28,630,000 and sample_set AG1000G-BF-A, and plots the coverage data for samples AB0086-Cx, AB0095-Cx and AB0123-C. Once you have changed the code, run those cells again. How many CNVs are found in each of those samples? How many extra copies are associated with the CNV in each sample?
Now go to the “Load the per-gene modal copy number data” section. Change the code again to focus on chromosome 3R and sample set AG1000G-BF-A. Create a heatmap of copy number for the genes in the Gste cluster. Which genes are found to have increased copy number in this sample set? Does what you see fit with the copy number plots that you made above for samples AB0086-Cx, AB0095-Cx and AB0123-C? Hint: here are the AGAP numbers for those genes:
gste_genes = dict(AGAP009195='Gste1',
AGAP009194='Gste2',
AGAP009197='Gste3',
AGAP009193='Gste4',
AGAP009192='Gste5',
AGAP009191='Gste6',
AGAP009196='Gste7')
Try again with samples AA0100-C and AA0135-C from sample set AG1000G-GH, looking again at Gste genes. How does that CNV compare with the one you found above? Which genes are now found to have increased copy number?
Français#
Ouvrir ce notebook dans Google Colab et l’exécuter vous-même du début à la fin. Indice: cliquer sur l’icône fusée () et sélectionner “Colab” dans le menu déroulant. Ensuite, cliquer sur le menu “Edit” et choisir “Clear all outputs” avant de faire démarrer le code dans les cellules. Confirmez que le notebook ne produit pas d’erreure, et assurez-vous d’avoir bien compris les résultats.
Retourner à la section “Load the CNV HMM data”. Changer le code pour qu’il analyse le chromosome 3R de la position 28,570,000 à 28,630,000 et le jeu de données AG1000G-BF-A, et qu’il trace le “coverage” pour les échantillons AB0086-Cx, AB0095-Cx et AB0123-C. Une fois le code changé, relancer l’analyse de ces cellules. Combien de CNVs se retrouvent dans chacun de ces échantillons? Combien de copies supplémentaires trouve-t-on du CNV dans chaque échantillon?
Maintenant, se diriger vers la section “Load the per-gene modal copy number data”. Changer une fois de plus le code pour qu’il se focalise sur le chromosome 3R et le jeu de données AG1000G-BF-A. Créer une “heatmap” du nombre de copies pour les gènes dans le groupe “Gste”. Quels gènes ont un nombre de copies élevé dans ce jeu de données? Est-ce que cela correspond aux traces de “coverage” que vous avez créées ci-dessus pour les échantillons AB0086-Cx, AB0095-Cx et AB0123-C Indice: voici les codes AGAP pour ces gènes:
gste_genes = dict(AGAP009195='Gste1',
AGAP009194='Gste2',
AGAP009197='Gste3',
AGAP009193='Gste4',
AGAP009192='Gste5',
AGAP009191='Gste6',
AGAP009196='Gste7')
Relancer l’analyse des gènes Gste, cette fois avec les échantillons AA0100-C and AA0135-C du jeu de données AG1000G-GH. Comparer le CNV que vous découvrez avec celui trouvé dans le AG1000G-BF-A. Quels gènes voient maintenant leur nombre de copies augmenter?
